A major development has been making the rounds recently: Apple’s “Mobile Pulse Rate Atrial Fibrillation Indication Recording Software” (i.e., the Apple Watch Atrial Fibrillation History Feature) officially obtained an NMPA imported Class II medical device registration certificate on December 26, 2025, under certificate number 国械注进20252210606. This approval does not cover the entire watch; it applies specifically to the atrial fibrillation monitoring software feature, which is designed for patients aged 22 and above with a confirmed diagnosis of atrial fibrillation. It can retrospectively estimate the proportion of time spent in atrial fibrillation episodes and correlate those findings with lifestyle data for analysis. Apple officially responded that it is following the formal regulatory process and expects the feature to be launched for use in China soon.
As a long-time professional in the medical device sector, this immediately caught my attention. Since securing FDA clearance for its ECG feature in 2018, Apple has built significant momentum in wearable medical technology, but the domestic Chinese market has long been constrained by the NMPA threshold. Now that it has finally cleared that barrier, it signals that major companies are accelerating the transition of consumer electronics into medical devices. For industry practitioners, this is a clear sign: wearables are no longer just “health trackers,” but tools that can legitimately enter clinical support scenarios. Consider the implications: atrial fibrillation patients could monitor AF burden (the proportion of time spent in episodes) in daily life without frequent hospital visits, greatly improving patient compliance and data continuity. That said, it is too early to be complacent. The bar for Class II registration is not low—from clinical validation to algorithm reliability, every aspect must be robust.

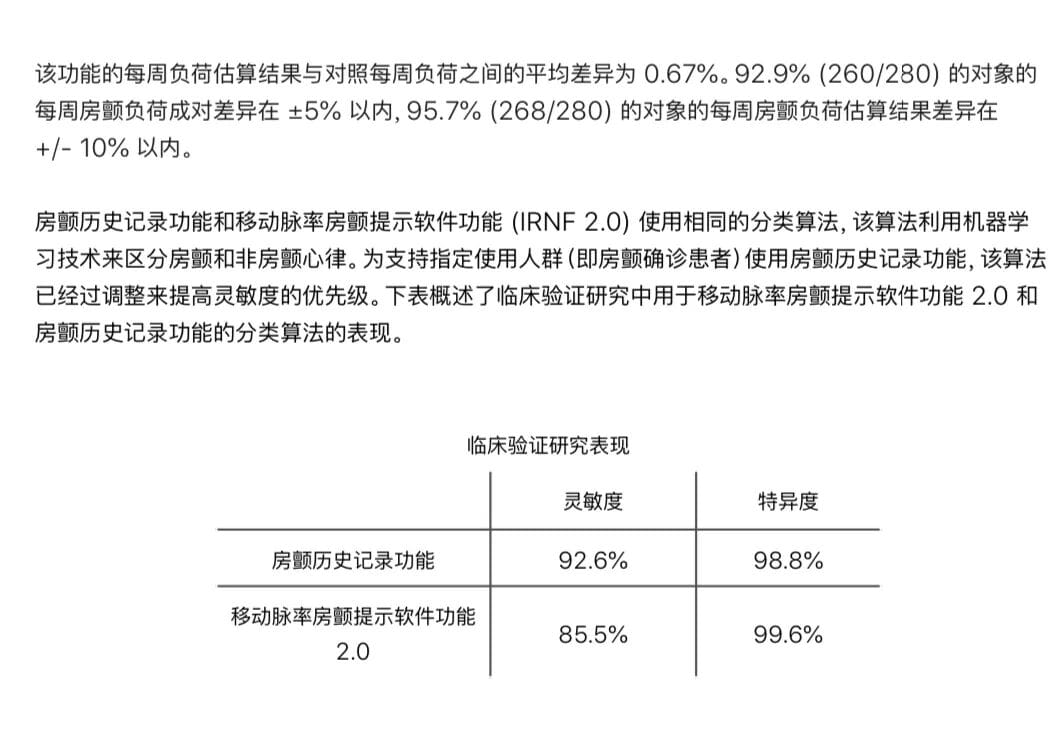
From a market perspective, this will intensify competition in China’s wearable medical device segment. Huawei, OPPO, and others obtained similar ECG approvals several years ago, and now that Apple is following suit, sales of this product category are expected to double again in 2026. However, the challenges are equally clear: the NMPA is imposing increasingly stringent requirements on algorithm bias control and data representativeness, and retrospective data alone is often insufficient for regulatory submission approval. Sponsors typically need to strengthen the evidence package with prospective clinical studies or RWD (real-world data). For example, when we previously supported a domestic smart wristband company with its Class II registration, we found that the algorithm lacked sufficient sensitivity/specificity data in Asian populations, which led to several rounds of deficiency letter response. Ultimately, we succeeded by adopting a hybrid evidence strategy (a small-sample RCT plus multicenter RWD), shortening the timeline by six months.
If you are a sponsor or startup team focused on similar AI-enabled wearable projects, the key takeaway is this: do not focus solely on technological innovation; you need to establish the regulatory pathway early. The core pain points in NMPA Class II registration lie in the Clinical Evaluation Report (CER) and software validation. For chronic disease monitoring functions in particular, you must demonstrate that the feature is not merely a “notification,” but a reliable “record.” The opportunity is that policy is encouraging digital transformation, and support tools for high-incidence conditions such as atrial fibrillation have enormous market potential. It is estimated that there are more than 10 million atrial fibrillation patients in China, and demand for routine monitoring is surging.
