🔎What Is EUDAMED
EUDAMED
Regulation (EU) 2017/745 (MDR) and related regulations have established a more stringent and transparent regulatory framework for EU medical devices and IVDR (in vitro diagnostic devices). Correspondingly,European Database on Medical Devices(EUDAMED)is designed to be the system’s “digital hub” — integratingmanufacturer / authorized representative / importer identity、device registration (UDI) / device information、Notified Bodies and certificates、market surveillance、post-market vigilance、clinical investigationsand other links, enablingfull-process traceability。
EUDAMED contains 6 modules:
“Actor Registration”
“Device / UDI Registration”
“Notified Bodies & Certificates”
“Market Surveillance”
“Post-market Surveillance & Vigilance”
“Clinical Investigation / Performance Studies”
Over the past few years, these modules have been rolled out gradually and were allowed only“voluntary use”;but as regulatory implementation advances, compliance will become mandatory。
Key timeline
EUDAMED
According to the notice issued by the European Commission on November 27, 2025,the first four EUDAMED modules have been confirmed to meet functional requirements,and have been published in the Official Journal of the European Union (OJEU)。

This means that, as of May 28, 2026 the following four modules will be mandatory:
Actor Registration
UDI/Devices Registration
Notified Bodies & Certificates
Market Surveillance
For medical device companies (manufacturers / authorized representatives / importers / system and procedure pack producers, etc.),this is no longer “optional” — it isa mandatory requirement for entering or continuing to sell products on the EU marketa hard compliance threshold。
Therefore, for companies seeking to export to Europe or legally sell medical device / IVD products in Europe, the period from now until 2026-05-28 is the last opportunity to prepare during the “transition period”.
What this means for medical device companies exporting to the EU
EUDAMED
Higher compliance thresholdIf Actor registration + UDI/device registration + NB/certificate registration + market surveillance information filing are not completed on time,this may directly affect CE certificate issuance, product export, placing on the market, and sales。
Full-lifecycle oversight: EUDAMED connects the entire chain from“who manufactures / who serves as the authorized representative / who imports + what the product is + Notified Body + certificates + post-market surveillance / vigilance / reporting ” across the full chain, with transparency and traceability significantly improved compared with the past.
Higher administrative compliance risk: Failure to submit required information through EUDAMED in accordance with the rules may lead to market access barriers, regulatory penalties, reputational damage, and more.
Limited time window:With only ~6 months remaining until 2026-05-28, companies that have not yet started preparing need to initiate compliance actions immediately.
For Chinese medical device companies, this means: if you plan to enter or continue in the EU market through EU compliance / CE certification / EU market access,you must make EUDAMED registration / data remediation / UDI system integration / coordination with your Notified Body / product documentation preparation immediate priorities.
Recommended Actions - Five Key Tasks
EUDAMED
Applicable to: manufacturers (including Chinese manufacturers), EU authorized representatives, importers, and system and procedure pack producers.
What does the company need to do? (Submission can be completed in 10–20 minutes)
Step 1: Prepare the materials
Business license
Manufacturer / EU Authorized Representative information
PRRC information (Person Responsible for Regulatory Compliance)
Company address and contact person
Step 2: Submit Actor registration
Log in to EUDAMED → Actor module → upload the documents.
Step 3: Competent authority review (3–10 days)
For non-EU companies, the review is conducted by the Member State where the EU Authorized Representative is located .
Step 4: Obtain the SRN
This is the unique identifier for all subsequent modules
② Complete UDI / device registration (the most time-consuming task)
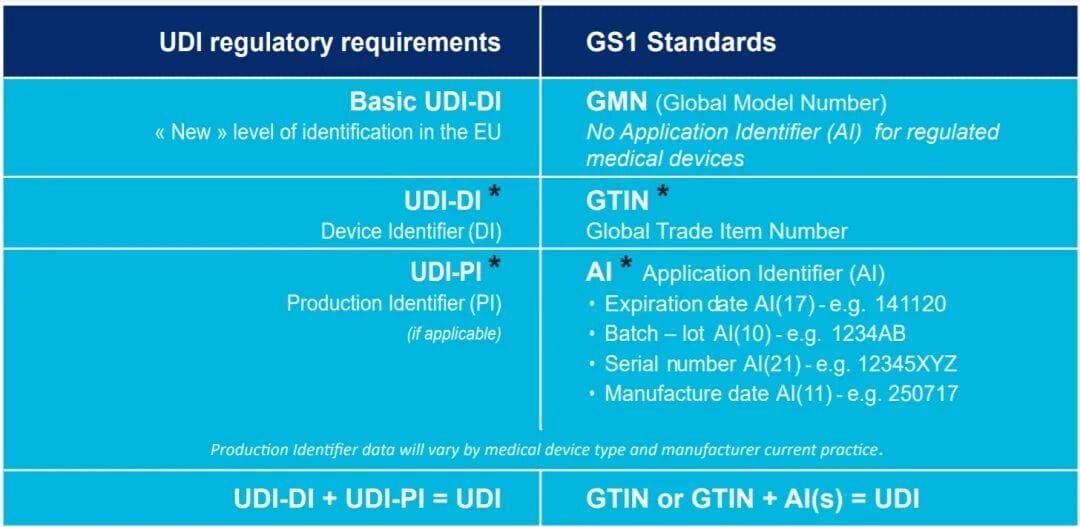
This is the step companies are most likely to underestimate
Step 1: Compile a product line list (mandatory)
Create an Excel sheet with the following fields:
Model
Risk class
EMDN code
Basic UDI-DI
UDI-DI
Packaging level
Step 2: Apply for / generate UDI
Via GS1 / HIBCC.
Step 3: Enter data in the EUDAMED Device module
Enter the following required fields:
Basic UDI-DI
UDI-DI
EMDN
Intended purpose
Packaging information
Manufacturer information
Step 4: Submit and retain records
③ NB certificate registration (must be uploaded by the Notified Body)
Actions required from the company
Step 1: Confirm the certificates cover the Basic UDI-DI
Otherwise, the NB cannot upload them.
Step 2: Notify the NB to upload the certificates on time
The company cannot upload them itself; this is a common misconception among many Chinese companies.
Step 3: Confirm within the system
Confirm that the certificate fields uploaded by the NB are correct.
Step 4: Regularly check certificate validity
Renewals and changes must all be synchronized.
④ Establish an internal EUDAMED update process (the most critical step once use becomes mandatory)
The company must establish 4 types of process:
1. Product change synchronization process
Changes to models, packaging, labeling, or manufacturing address → must be updated.
2. Certificate status update process
Renewals, changes, and scope extensions must be synchronized promptly.
3. PMS / Vigilance (post-market) process
Adverse events and Field Safety Corrective Actions (FSCA) must comply with the requirements of MDR Article 87–100.
4. Quarterly data review process
Ensure consistency across UDI, certificate, and product data.
⑤ Appoint a PRRC + establish an EUDAMED project timeline
This is an area many companies overlook
Responsibilities of the PRRC (Person Responsible for Regulatory Compliance) (MDR Article 15)
Review all EUDAMED data
Ensure products comply with MDR requirements
Oversee PMS / Vigilance data
Recommended project timeline (applicable to 2025–2026)
2025-12: Complete the SRN (Actor module) + complete the master product line list 2026-01: Generate the Basic UDI-DI / UDI-DI + prepare EUDAMED fields 2026-02: Complete ≥50% UDI / Device entry 2026-03: Complete upload of all NB certificates + company confirmation 2026-04: Internal data review (full verification of UDI, Device, and Certificates) 2026-05: Final PRRC review → full-module check → 5/28 mandatory go-live
The earlier you prepare, the less likely you are to miss the deadline and fail.
After May 28, 2026, all companies exporting to the EU must comply.

