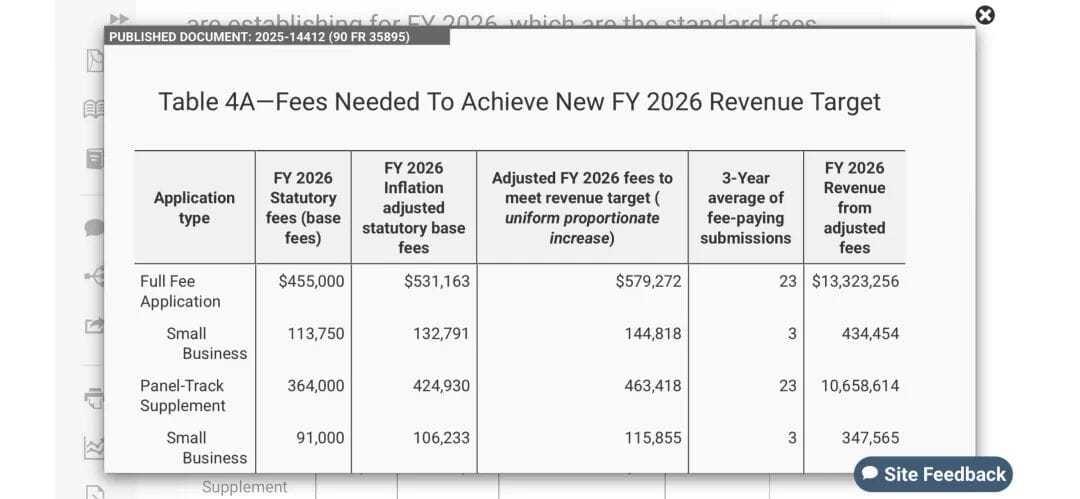
FDA recently published the FY 2026 medical device user fee schedule in the Federal Register. The annual establishment registration fee is set at US$11,423, slightly higher than last year. Application fees have also been adjusted accordingly: the standard PMA fee is US$579,272, with a small business rate of US$144,818; the standard 510(k) fee is US$26,067, with a small business rate of US$6,517. In simple terms, for an SME involving establishment registration and one 510(k) submission, the additional cost could increase by several thousand dollars. 
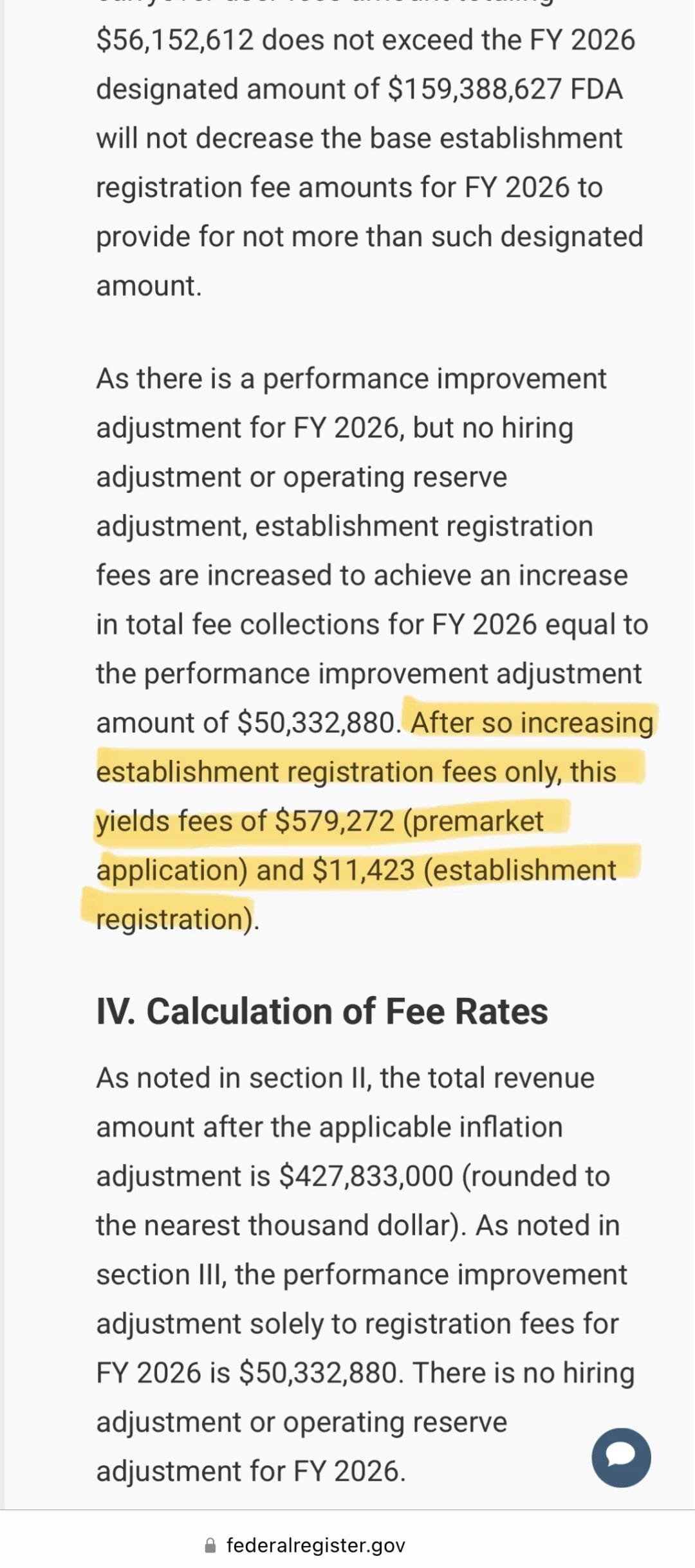
Export-oriented companies in China have noted that this round of changes will add to the burden, especially for smaller manufacturers, where compliance costs already account for a meaningful share of expenses. FDA’s total target revenue has now been adjusted to approximately US$428 million (US$366 million base, increased for inflation), plus US$50.33 million from performance improvement adjustments, bringing total expected revenue to nearly US$478 million. These funds are mainly used for reviewer salaries, facility maintenance, and operations to support faster and more efficient device review.  But for companies, this also means a higher threshold for entering the U.S. market.
Compared with NMPA fees, the gap is significant. The NMPA initial registration fee for imported Class II devices is RMB 210,900 (about US$27,000), and for Class III devices RMB 308,800 (about US$39,000). For change registration, the fee is RMB 42,000 for Class II and RMB 50,400 for Class III, with a five-year renewal fee of RMB 40,800. There is no annual establishment fee. In terms of timelines, NMPA Class II typically takes 16-24 months and Class III 24-36 months, with overall costs that are easier to plan and control. FDA PMA costs more than 10 times as much as NMPA Class III registration, and there is also an annual registration fee. Of course, the U.S. market is larger and offers higher returns; the key is how to balance the upfront investment.
Based on experience, the following points may be helpful:
• When budgeting, assess the product classification carefully. Fee increases have a greater impact on high-risk Class III devices, while many low-risk Class I/II devices benefit from exemptions. Small businesses (revenue < US$100 million) can apply for reduced fees and save 75%; if revenue < US$30 million, the first PMA may be fully waived.
• During the submission process, minimize requests for additional information. QMSR takes effect in February 2026, and alignment with ISO 13485 can improve efficiency.
• From a resource perspective, the De Novo pathway is suitable for innovative devices and comes with lower fees. It is worth reviewing FDA guidance or insights shared by domestic industry associations.
• For market strategy, if U.S. costs are too high, there are ongoing discussions around simplification under the EU MDR, or companies may choose to focus on the domestic market, where NMPA costs are comparatively manageable.
This is a continuation of the MDUFA V framework, which is reviewed every five years. Some companies have adapted by focusing on high-value products or optimizing their value chain. If you have relevant experience, feel free to share it in the comments.
