Do you know about the update? Do you know the deadline? Do you have an adaptation plan?
1. Start with the conclusion: FDA explicitly states a “transition period”; this is not a rumor
Stop relying on secondhand summaries; FDA has clearly recorded it in the Recognized Consensus Standards database:
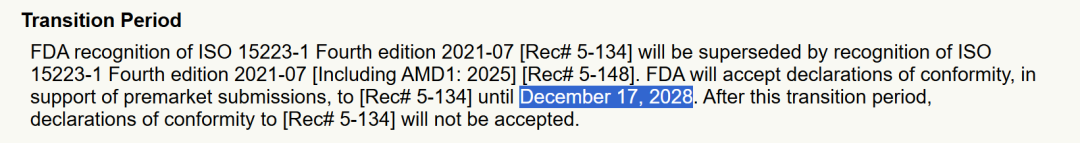
ISO 15223-1, 4th edition, 2021-07, including AMD1:2025 (Rec# 5-148), entry date 2025-12-22.
Declarations of conformity to the previously recognized version (Rec# 5-134) may be accepted until 2028-12-17; after that, declarations to the previous version will no longer be accepted。
In one sentence: you can transition on your own timeline, but you cannot act as if this never happened.
2. What changed in AMD1:2025, and why has it become an “audit topic”?
Many teams’ first reaction is: they’re just symbols—update the icons and you’re done.
But the signal in this update is clearer: the standard is shifting“representative information/symbol expression” away from region-specific usage and toward more general expression.
ISO’s key descriptions of AMD1 include:
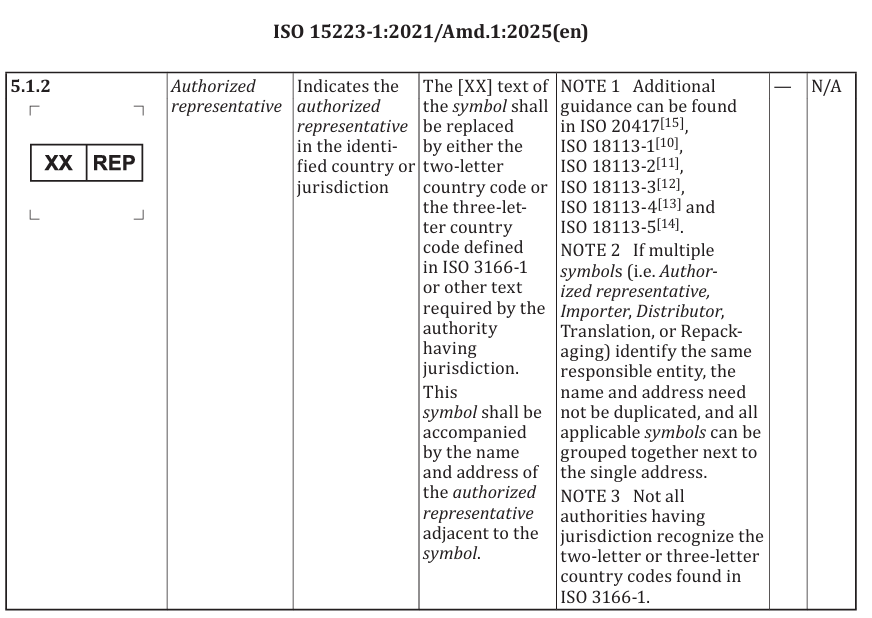
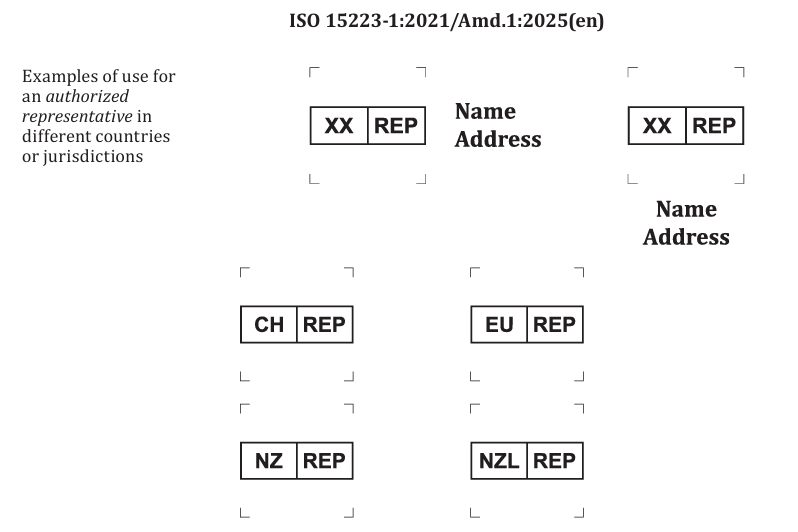
new defined terms related to the “authorized representative”
revision of the representative symbol that originally leaned toward “EC-REP” semantics, so it is no longer tied to a specific region
This naturally triggers auditors to ask:
Does your labeling strategy still reflect the old interpretation?
How is representative information managed consistently across multiple jurisdictions?
Do you have a documented chain of change evidence?
3. Why you may not be “required to change immediately” now, while auditors will “ask immediately”
Becausethe transition period protects the implementation window, whereasaudits assessmanagement capability。
If you think it through from the auditor’s perspective, it becomes clear—they will not ask “have you changed it yet,” they will ask a three-part series:
1) Are you aware that the standard has been updated?
2) Do you know the FDA transition-period deadline? (2028-12-17)
3) Do you have an adaptation plan? Who is responsible? When will it be completed? Where is the evidence?
If your answers are vague, it will escalate into a system-level issue:
Is your standard-monitoring mechanism effective?
Is document control fully closed-loop?
Are the label, IFU, and technical documentation consistent?
4 If you do not know and have no plan, what are the practical risks?
I suggest you put the following sentence into the department meeting PPT:
“The longer the transition period,the easier it is to mistakenly assume it can be ignored。”
The common risk is not that “a single symbol triggers a major finding,” but the chain reaction that follows:
Questions about whether your regulatory surveillance/standards list update mechanism has become ineffective
Being required to provide additional impact assessments, change records, and training evidence
A last-minute switchover close to the deadline, with supply chain, packaging, and registration changes all bottlenecking at once
5 Here is a very safe answer to use during an audit (recommended)
Q: Are you aware of the ISO 15223-1 update?
A: Yes. We have included ISO 15223-1:2021 (including AMD1:2025) in our applicable standards list and completed the impact assessment and implementation plan.
Q: When does the FDA transition period end?
A: FDA will accept declarations of conformity referencing the previously recognized version, Rec# 5-134, until 2028-12-17; after that, declarations referencing the old version will no longer be accepted.
Q: When will you complete the transition?
A: We are executing under the principle of “prioritizing new/changed products and transitioning existing products in batches,” and we have set our latest internal deadline earlier than 2028-12-17 to avoid a concentrated last-minute changeover.
6 The least burdensome approach for companies: build one auditable evidence chain and resolve the issue properly in one go
When we support clients with this type of transition, our goal is never just to “draw the symbols correctly,” but to have the full evidence chain ready before the auditor asks for it.
0-2 weeks: turn “we know” into documentation
Update the applicable standards list: include ISO 15223-1:2021 (including AMD1:2025)
Write a 1-page impact assessment: which products and labeling materials are affected, and whether a design change is triggered
2-6 weeks: turn the “impact” into a plan
SymbolsGap checklist(SKU - material - country - switch-over point)
SetTransition strategy: New products/changes adopt the new version first; existing stock switches by batch
Set an internal latestswitch-over point, do not wait until 2028-12-17
6-12 weeks: turn the “plan” into an evidence trail
Document control: version change records, approvals, release, training
Risk management: reflect the label-change assessment conclusions and rationale in the RMF
Supply chain: packaging material consumption / dual-version coexistence management to prevent mixed packing