Engineering solutions to clinical pain points, application prospects, and commercialization perspectives (Science Advances, 10.1126/sciadv.adn5195 / arXiv:2312.12581)
Overview:Constrained by traditional receive coils that are bulky, rigid, and heavily wired, MRI has long faced trade-offs among patient comfort, positioning flexibility, and SNR performance.This article examines the Boston University team’s proposed“wireless, ultralight, coaxially shielded coils (CCR)”differentform-factorapplications:spine-conforming, wrist-clasp, and fingerring-type coils (CCR)as well as their array-based metamaterial approach, showing how these engineering designs address clinical pain points and assessing their potential application scenarios and commercial value.
“Wireless + customizable +and body-conforming—this may be theMRIfuture form of coils.”
“CCRTurning coils from‘equipment’into‘wearables’。”
“What engineers are doing is turning complex electromagnetics into a flexible line.”
“In the future, the moment a patient puts on the coil, the scan may already have begun.”
1. What exactly is novel about this study?
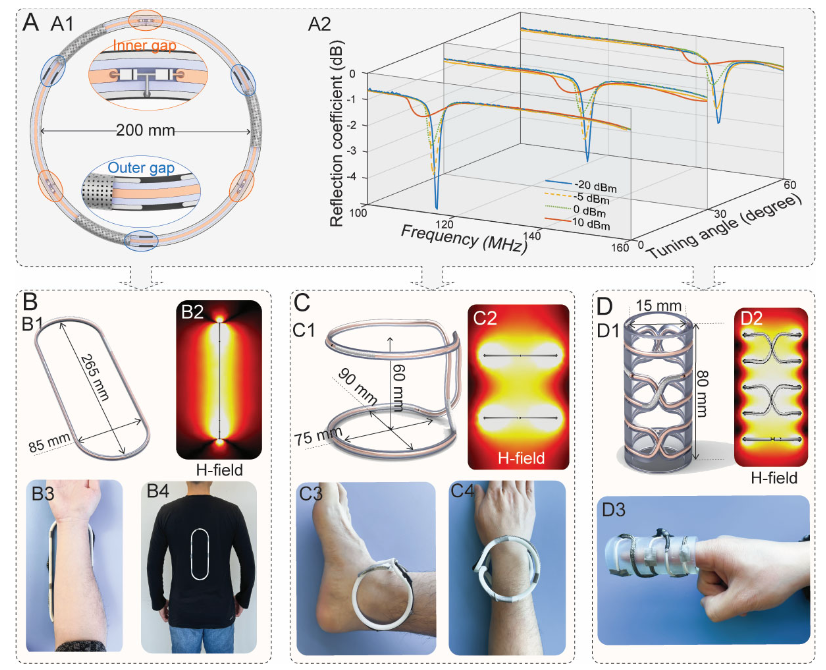
1)Coaxial shielding structure + multi-slot capacitance:By creating slots in the inner/and outer conductors of the coaxial cable, opposing gaps create a strongly confined electric field and magnetically dominant resonant behavior, avoiding the electrically coupled noise of conventional coils to the sample and improving QQ factor andSNR potential.
2)“Tunable, customizable, and adaptive” in one:A rotatable“tuning sleeve” enables~30–60 MHz frequency fine-tuning over a range; the multi-slot design allows the diameter/ and form factor to be customized while the frequency remains more stable; placingPIN diodes in parallel across the gaps enables“ enhanced low-power reception and decoupling during high-power transmission” as an adaptive response——It automatically detunes during transmission and strengthens the magnetic field during reception.
3)Truly“wireless, ultralight, conformable” form-factor freedom:CCRIt can be bent and twisted into conformal bands for the spine and arm/wrist/Snap-on ankle coils, finger ring coils, and similar designs significantly shorten“coil-to-—anatomy ”distance, deliveringhigher local sensitivitySNR and comfort。
4)The array itself acts as a metamaterial,:using two turnsCCRas the unit, assembled laterally (H-) or longitudinally (V-) to form“a scalable field of view” metamaterial array; in the proper operating mode, currents in each unit flow in the same direction, the magnetic fields superimpose with enhanced localization, while maintaining unit-level sensitivity.
II. Which clinical pain points does it address?
• Coils are bulky/and rigid, with poor positioning flexibility and comfort, especially for small anatomies and complex patient positioning;
• A single scanner often requires multiple“anatomy-specific coils”, resulting in high hardware investment and management costs;
• Cable-related routing and trap-circuit burdens affect workflow efficiency and patient experience;
• Conventional array sensitivity enhancement and field-of-view expansion often require a choice between sensitivity and coverage“—one or the other”。
III. Key data:SNRMeasured comparisons and sequence compatibility
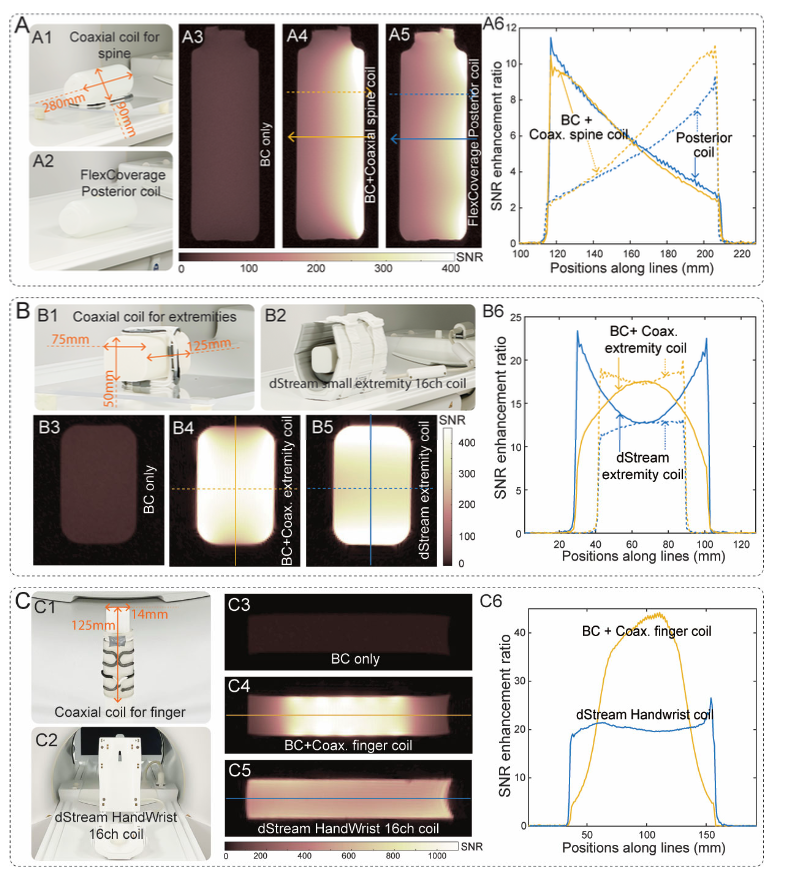
• Phantom experiments: compared with the body transmit coil (birdcage, BC), the spine-conformal CCRdelivers approximately 10-foldSNR improvement; the conformal “small-body-part coil” yields CCRapproximately 17.5-fold;“finger coil”approximately 44-fold. Compared with Philips commercial 16-channel small-body-part coil/ and wrist/finger coil, CCRhigher SNR。
• Array (metamaterial) experiments: while expanding coverage, SNRit was not diluted and showed a clear improvement over comparable commercial coils.
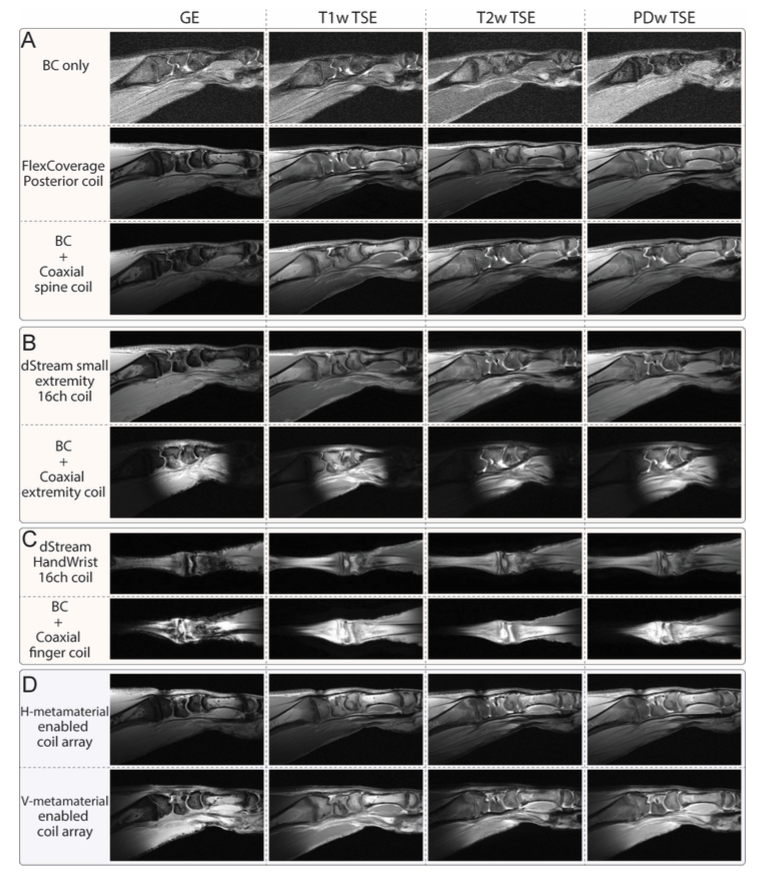
• Ex vivo samples: pig leg/ and chicken claw under GE、T1w/T2w/PDw TSE and other commonly used clinical sequences, CCRmaintained good image quality, confirming sequence compatibility.
IV. Engineering principles: why it can be “powerful, lightweight, and wireless”?
• Primarily magnetic response: the coaxial structure causes the currents on the inner sides of the inner and outer conductors to cancel each other, so only the current on the outer surface of the outer conductor contributes to the magnetic field, suppressing unnecessary electric coupling and reducing the introduction of sample noise.
• Strong electric-field confinement: the electric field is mainly confined to the gaps and dielectric layer, reducing harmful coupling with the human body and improvingSNR。
• Adaptive decoupling:PINduring high-power transmission, the diodes effectively increase capacitance and alter resonance, causing the coil to“detune and become inactive”; when receiving microwatt-level signals, resonance is restored and enhancedB1-, equivalent to“automatic safety+automatic sensitivity enhancement”。
• form-factor freedom + Tunable frequency: frequency shifts caused by deformation are compensated by the“tuning sleeve”; multiple gaps make resonance more stable across different diameters, facilitating rapid customization of anatomy-specific form factors.
V. Potential clinical application landscape (by scenario)
A. High-resolution imaging for small body parts: fingers/wrist/ankle/toes, etc., pursuing ultra-near-field performance and ultimateSNR;
B. Difficult-to-position populations: patients with pain, postoperative immobilization, or pediatric patients, with emphasis on comfort and rapid positioning;
C. spine/Full-length extremities: byH-/V-extending coverage with an array configuration while balancing sensitivity and field of view;
D. Outpatient and mobile use: ultra-light wireless design streamlines workflows and may improve throughput and user experience;
E. Resource-constrained settings: extremely low manufacturing cost and customizability may lowerMRIthe barrier to use.
VI. Comparative analysis with traditional/flexible/modular coils
Dimension | Conventional surface/phased-array | Flexible/fabric/liquid-metal coils | This study's CCR unit | CCR metamaterial array |
Comfort/fit | General/rigid | Good | Excellent (snap-on/fit) | Excellent (expanded coverage while remaining conformal) |
SNR | High (when close-fitting) | Comparable | Locally higher | Maintains high performance even with expanded coverageSNR |
Cable burden | High | Medium-high | Low (wireless) | Low (wireless) |
Positioning efficiency | Limited | Better | Excellent | Excellent |
Cost | High (multiple dedicated sets) | Medium-high | Low (can be manufactured at low cost) | Low (expand as needed) |
Parallel imaging | Mature | Mature | May be limited (requires further validation) | May be limited (requires further validation) |
VII. Commercialization and ecosystem impact assessment
• Value proposition: reduce costs and improve efficiency without sacrificing image quality——Increase throughput, shorten setup time, reduce coil inventory and maintenance, and improve patient experience.
• Target customers: public hospital imaging departments, musculoskeletal and sports medicine centers, pediatric institutions, and outpatient/emergency departments/mobileMRI; developing-country or primary-care markets.
• Business model: “unit+arrays”-based modular sales; provide anatomy-specific “rapid customization kits” (diameter/, shape/, and sleeve presetting); with supporting consumables and training services.
• Cost structure: coaxial materials and3D low-cost printed support components; the core lies in reliable RF component manufacturing, assembly consistency, and clinical-grade testing and certification.
• Competitive barriers: magnetic-response-basedCCR physical mechanisms, adaptive decoupling, electromagnetic safety boundaries, and clinicalSNR evidence accumulation.
8. Risks, Compliance, and Implementation Challenges
• Parallel imaging/ channel count: wireless design may limit parallel acquisition efficiency and requires joint tuning and validation with the scanner ecosystem;
• Compatibility: different manufacturers (1.5T/3T/7T) system differences, sequence diversity, andSAR regulatory requirements;
• Durability and cleaning: repeated bending/Mechanical durability of the snap-fit structure, disinfection workflow, and material biocompatibility;
• Regulatory pathway:As an accessoryRFreceiver device, it must undergoIECRF safety testing under applicable standards,EMCand clinical performance validation;
• Market education: the shift from“dedicated multi-coil configurations”to“modular+arrays”also brings mindset changes and training costs.
Conclusion
CCRTurning“the ideal that coils shouldconform closely to the body, remain flexible, and also be wireless”into an engineering-ready solution. If its stable benefits continue to be validated in parallel imaging and large-scale clinical use, this new paradigm of“customizable units + and scalable arrays”may reshapeMRIthe product logic and commercial boundaries of coils.
Follow Clinsota for more technology insights
