 Among these, the Article 61(10) performance-based clinical evaluation pathway is an especially controversial topic, with differing views across the industry. Today, let us take a closer look at the Article 61(10) performance-based clinical evaluation pathway, compare perspectives while respecting differences, and hopefully provide some useful ideas and approaches for those who need them.
Among these, the Article 61(10) performance-based clinical evaluation pathway is an especially controversial topic, with differing views across the industry. Today, let us take a closer look at the Article 61(10) performance-based clinical evaluation pathway, compare perspectives while respecting differences, and hopefully provide some useful ideas and approaches for those who need them.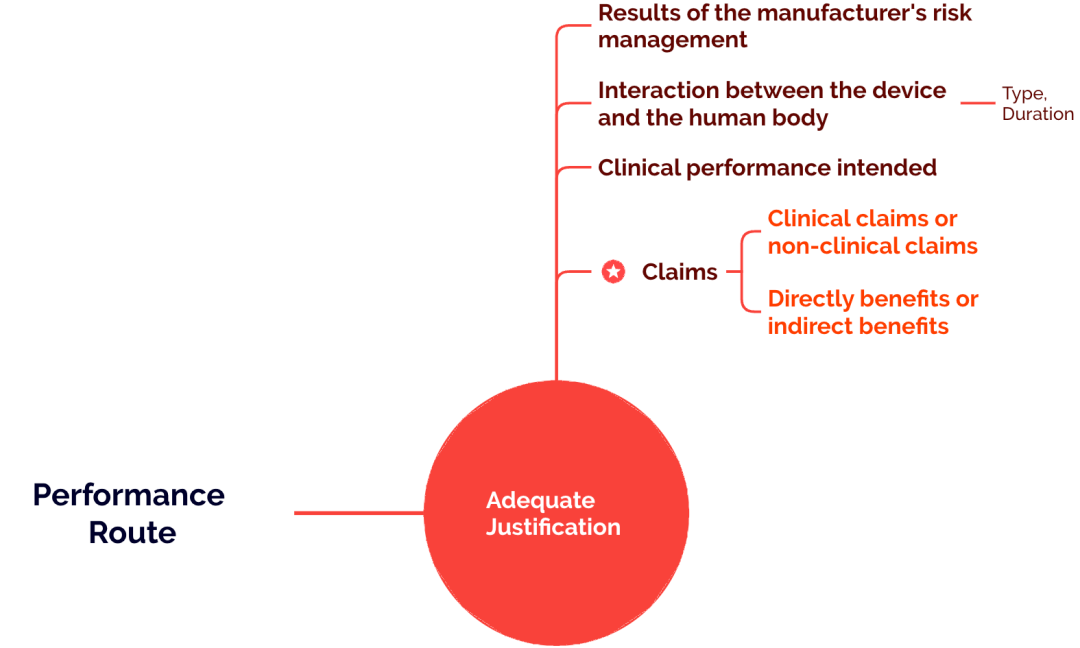
1. What is the origin of Article 61(10), and what are its core considerations?
2. Which devices may follow Article 61(10)?
3. What data can support the performance-based pathway?
4. What are the advantages and limitations of the performance-based pathway?
1. The origin and core considerations of Article 61(10)
According to Article 61(10) under MDR Chapter VI, Clinical Evaluation, commonly referred to in the industry as Article 61(10), the original intent is that if it is considered that demonstrating the device’s safety and performance throughclinical data is not appropriate, then the manufacturer may consider completing the clinical evaluation through a performance-based pathway, but the manufacturer must provide aAdequate Justification, which should be based on the following key considerations
the results of risk management
the nature of the device’s contact with the human body (mode of contact, duration of contact, and novelty of the interaction)
the manufacturer’s intended clinical performance
claims (especially clinically related claims)
Among these, the main core criterion—or“watershed”, in determining whether an EU clinical evaluation may follow the performance-based pathway is whether the target device has a direct clinical benefit claim(direct clinical benefits claims). This is the primary basis for judgement. Sometimes making this determination can be difficult. If you are uncertain, you may consider the issue from the following perspectives:
Device has direct influence on clinical outcomes or not?
The clinical benefits can be specified through measurable or not?
Positive impact on Patient-relevant healthe/ clinical outcome(s)?
The clinical benefit is direct OR indirect?
It should be noted that the prerequisite for considering the performance-based pathway is that the device must not be an implantable device or a Class III device (as such devices must undergo clinical investigation).
Now that we have discussed the rules, many of you may be even more curious about which devices can actually use the performance-based pathway to complete clinical evaluation. Let us therefore look at some current examples (different Notified Bodies may have different opinions). With continued practice by manufacturers and Notified Bodies across the industry, this understanding will likely continue to evolve.
2. Which devices can actually follow the Article 61(10) performance-based clinical evaluation pathway?
We all want clear answers on which devices can follow Article 61(10), so that we can use them as a reference in assessing whether our own devices may also qualify.
Before that, it must be emphasized that the Article 61(10) pathway is not an “excuse” or a “shortcut” for having no clinical data. For example: our device is simple and functions normally, with almost no complaints; no one would conduct dedicated studies on our device because it is only an auxiliary tool; or no relevant literature can be found through a clinical literature search. (Absence of evidence is not evidence of absence)
Rather, it is based on the actual design and intended purpose of the device, where it is genuinely considered that clinical data is not necessary to demonstrate the device’s safety and performance. For example, below is a summary of some devices that, based on current practical experience, may follow the performance-based clinical evaluation pathway:
Device category | Examples | Consideration factors |
Laboratory equipment | Blood refrigerators, laboratory balances, centrifuges | No direct patient contact and no clinical benefit |
Basic surgical instruments, someWETdevices | forceps, scissors, hemostatic forceps, retractors,low-risk devices such as wires, needles, clips, and connectors | Contact with the human body but no energy exchange; mature design |
Dental equipment | dental treatment units, cheek retractors, dental mirrors | Low risk, primarily structural and functional |
Auxiliary devices | Wheelchairs, walkers, patient positioning systems, surgical lights, medical ice packs, immobilization bandages, etc. | Primarily mechanical in structure, with no therapeutic function |
Partially standalone software | Ancillary software that does not directly affect diagnostic or treatment decisions | No diagnostic or therapeutic claims |
Passive measurement devices | Stethoscopes, blood glucose meters (non-diagnostic), thermometers | May be considered if no clinical benefit is claimed61(10) |
Device accessories | Accessories that do not directly affect the performance of the main device, such as catheter accessories and consumable accessories | No independent clinical claims; functional verification only |
Console-type devices | For example, some consoles used with balloon catheters only provide energy | Provides only energy, with no direct clinical benefit |
Medical consumables | Syringes, catheters(tube not catheter), auxiliary materials, ultrasound coupling gel, dental impression materials, non-implantable drainage tubes, cleaning agents or disinfectants, | No claims of direct clinical benefit |
Low-risk rehabilitation devices | Walkers, crutches, etc. | Functional claims only |
Low-risk monitoring devices | Temperature probes, etc. | Functional verification only |
Adjunctive diagnostic imaging devices | Provide medical images only | No direct diagnostic, therapeutic, or other clinically relevant claims |
3. If the performance pathway is feasible, what data can actually be used to demonstrate the safety and performance of your device?
Bench testing
Common specifications, Harmonized standards related test
Pre-clinical data
such as mechanical testing for strength and endurance, biological safety, usability, etc
Performance data
Verification&Validation, Volunteer test, animals test, Simulated use / animal / cadaveric testing involving healthcare professionals or other end users
PMS/Vigilance Data (If device has previously been marketed)
Include PMCF data
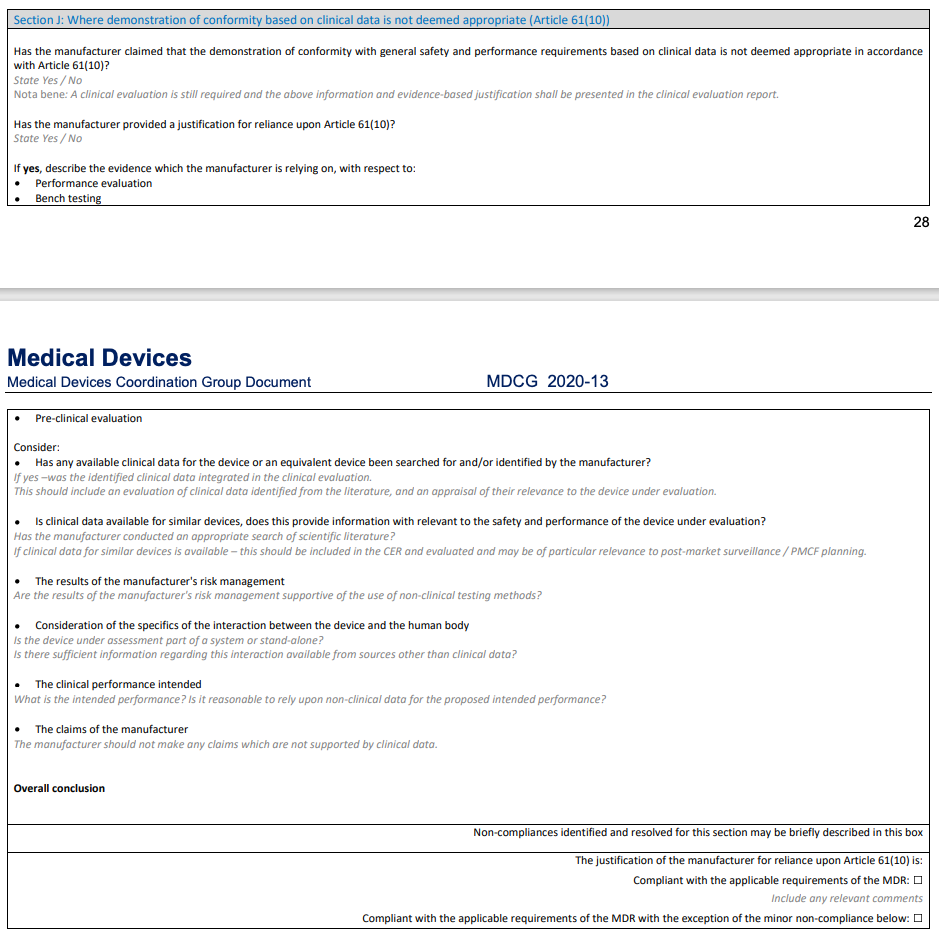
For practical implementation guidance, please refer to MDCG 2020-13 Section J (the requirements and checklist for clinical evaluation under Article 61(10)).
4. Why is the performance-based clinical evaluation pathway both an opportunity and a challenge?
Some readers may want to push back when they see this topic: WHAT?! What opportunities are left in an EU MDR CER? It feels like challenges everywhere, doesn’t it?!
Of course, this so-called opportunity is conditional. For the relatively low-risk devices mentioned above, where it is considered inappropriate to demonstrate the device’s safety and performance through clinical data, the performance-based clinical evaluation pathway is undoubtedly an opportunity. Since it is an opportunity, it comes with certain benefits. For example, exemption from a clinical investigation can bring significant compliance advantages and efficiency gains for low-risk medical devices. Its core value is reflected in three areas: better regulatory fit, reduced cost and shorter timelines, and faster market access for technologically mature products. At the same time, however, proving that your device is eligible for the performance-based clinical evaluation pathway often requires more rigorous analysis and planning, with sufficient justification and a coherent line of reasoning to demonstrate that the device qualifies. The real difficulty is that many companies are unable to make a scientifically sound judgment on their own (which often requires professional input from clinicians), while also being unable to convincingly demonstrate device eligibility to the Notified Body. That is why we have compiled some practical success cases and implementation experience today, in the hope of offering useful reference points or ideas to those who are still struggling with the performance-based clinical evaluation pathway.
It should also be emphasized that taking the performance-based pathway does not mean you can omit the CER. CEP, CER, PMCF, PSUR, and the other required documents are all still indispensable. Nor does performance-based clinical evaluation mean that literature searching and literature analysis can be skipped, or that clinical evidence cannot appear in the clinical evaluation report. Demonstration of State of the Art (SOTA) and GSPR conformity remains essential. For details, please refer to the guidance provided in MDCG 2020-13 Section J above.
Finally, I would also like to say something from the heart,Clinical evaluation strategy must come first, clinical evaluation is not merely a clinical compliance report, nor is it a report that simply claims what a device can do; rather, it mustdemonstrate what the device can do and how well it performsa process that spans the entire device lifecycle; it is strongly recommended to establish the device’s clinical evaluation strategy before R&D, rather than waiting until the product is fully developed and all required verifications or tests have been completed, and only then starting to “piece together” an “all-inclusive” Clinical Evaluation Report from a pile of data and documents. Clinical evaluation is an“opportunity to demonstrate its validity”!!! It must bea coherent, logical, and tightly connected “clinical narrative that can demonstrate your device is feasible~”
Finally, to stay on topic today, let me say a few more words about the challenges of the so-called performance-based clinical evaluation pathway. If a company or device has ample funding and is willing to invest heavily, it may also conduct a clinical trial to support market promotion. Therefore, if you follow the performance pathway, you cannot have explicit and genuine “clinical claims.” Some people may think this is unfavorable for product marketing and promotion. However, in my view, we should return to the essence of the device: if “clinical claims” exist, they exist; if they do not, they do not. Conducting a clinical trial will notbeyond the intended purpose create so-called “clinical claims.”
Last but not least, at this point some of you may raise a fundamental question: if I follow the performance-based clinical evaluation pathway, then where does the“soul” - Safety and performance parameters and acceptance criteria & SOTA of my overall clinical evaluation come from? That is an excellent question—stay tuned for the next discussion~
Reference


