4. How can we distinguish a true “clinical claim” from a false one?
2. What does the EU MDR require for the Clinical Claim in a CER?



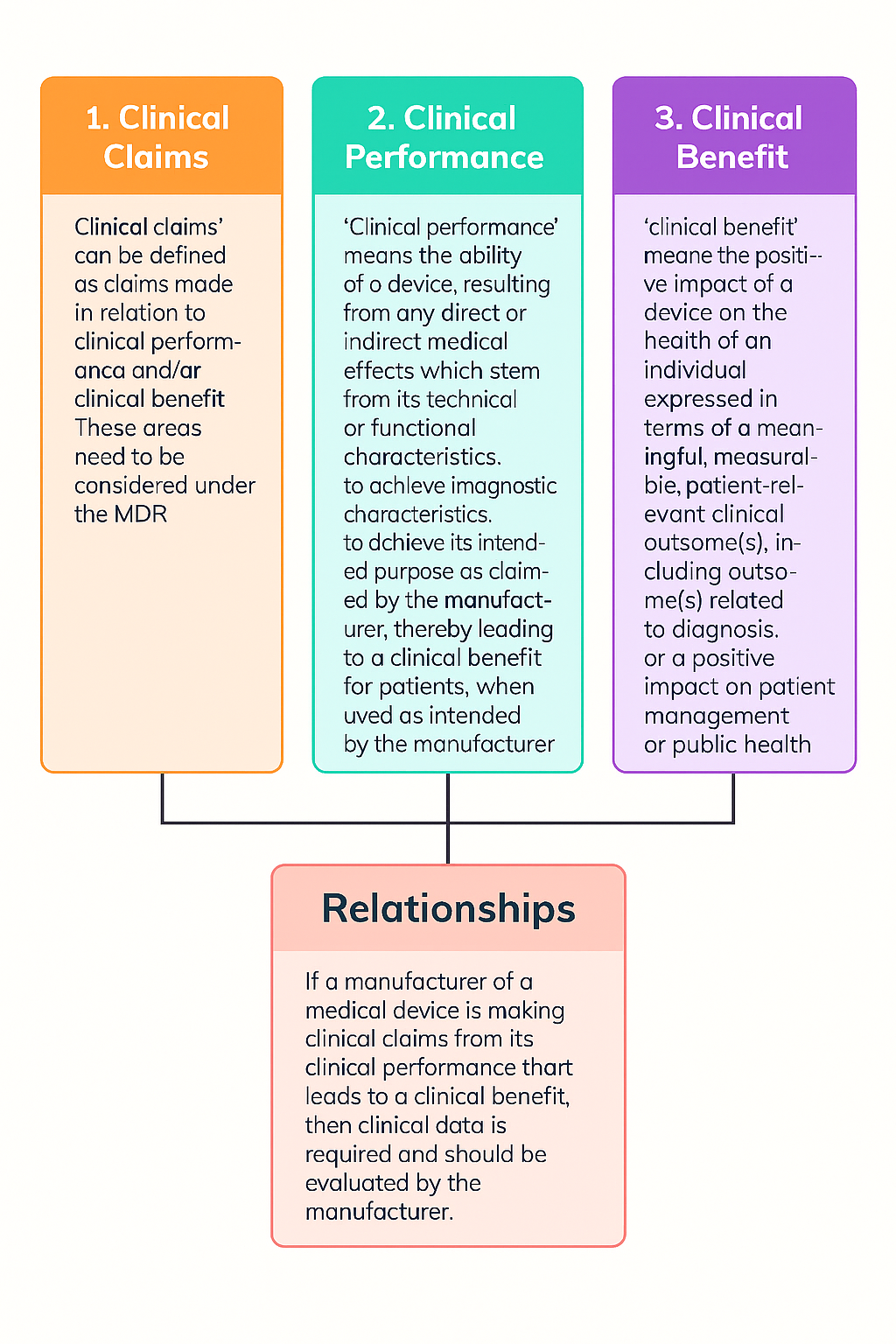
4. How do you assess whether a “Clinical Claim” is valid?
Before giving examples, one point should be clarified: the same device may have non-clinical claims or clinical claims; the key question is whether you can substantiate your claim with sufficient evidence.
A. Let us start with a very simple example. For a basic scalpel, if your clinical claim is that it cuts/dissects tissue to facilitate the procedure, then this is a non-clinical claim and can also be understood as a performance claim. The intended claim is only to achieve the intended function/capability/ability, and the patient benefit is only an indirect clinical benefit, because the ultimate success of the surgery, or the clinical outcomes, is not determined by the scalpel itself.
B. The same applies to a scalpel, but if the clinical claim is that it cuts/dissects tissue and can reduce procedure time by XX%, then such a claim is a clinical claim, because the scalpel affects procedure time and in turn affects patient clinical benefit (positive impact).
Based on this typical example, the real point is that when making a Clinical Claim, please “think twice” and consider the broader impact. We learned this the hard way: because a Clinical Claim went beyond the intended purpose, extensive remediation was required—revising the IFU, marketing materials, data sheets, and all related TD documents. The most difficult part is when the product has already been on the market for many years: how do you withdraw your TD for revision? Therefore, before making any claim, first set a clear objective for yourself—you must have sufficient evidence to support your claim, not merely assert it.
C. Here are two more examples:
A surgical suture is the first choice of 95% of physicians?
A CT device can provide CT images with higher contrast?
Please consider whether these two claims are Clinical Claims, and let me know your answer in the comments.
Finally, returning to the main topic, why do we saythat the Clinical Claim is both the “starting point” and the “end point” of the CER? There are two dimensions to this,From the perspective of device R&D and design, the original intent behind designing your device was not only to establish a presence in the market, but also to provide benefits. Those benefits may be for patients or users, and they may be direct or indirect. Therefore, the claim (whether a clinical claim or a non-clinical claim) is the“the original intent” or “the selling point”, so you want to design a device that matches the “selling point” you expect; the other is fromthe regulatory perspective, or, from a clinical evaluation perspective, precisely because you want to claim what your device can do, you need to mine sufficient evidence based on the claims in your intended purpose to support and substantiate those claims. So the claim you want to make is only the starting point, while the ultimate endpoint or focus of the CER still serves that claim; that is, you prove what your device can do or to what extent it can do it, and while it can achieve its intended purpose, you must also ensure an acceptableBenefits-Risk Ratiobenefit-risk balance—that is, patients should not incur harms greater than the benefits they receive; the above is whyThese are my personal thoughts on why a Clinical Claim is the “starting point” and “endpoint” of a CER. I hope I explained it clearly and that you found it helpful~

