On the morning of April 10, 2026, Shenzhen Clinsota Technology Consulting Co., Ltd. will appear at CMEF 2026 Spring, held at the National Exhibition and Convention Center in Shanghai, and has been invited to participate in the “2026 3rd Continuous Glucose Monitoring (CGM) Technology Seminar” and deliver a featured presentation at the conference:“Global Registration Strategies for CGM: Clinical Compliance Challenges and Solutions.”
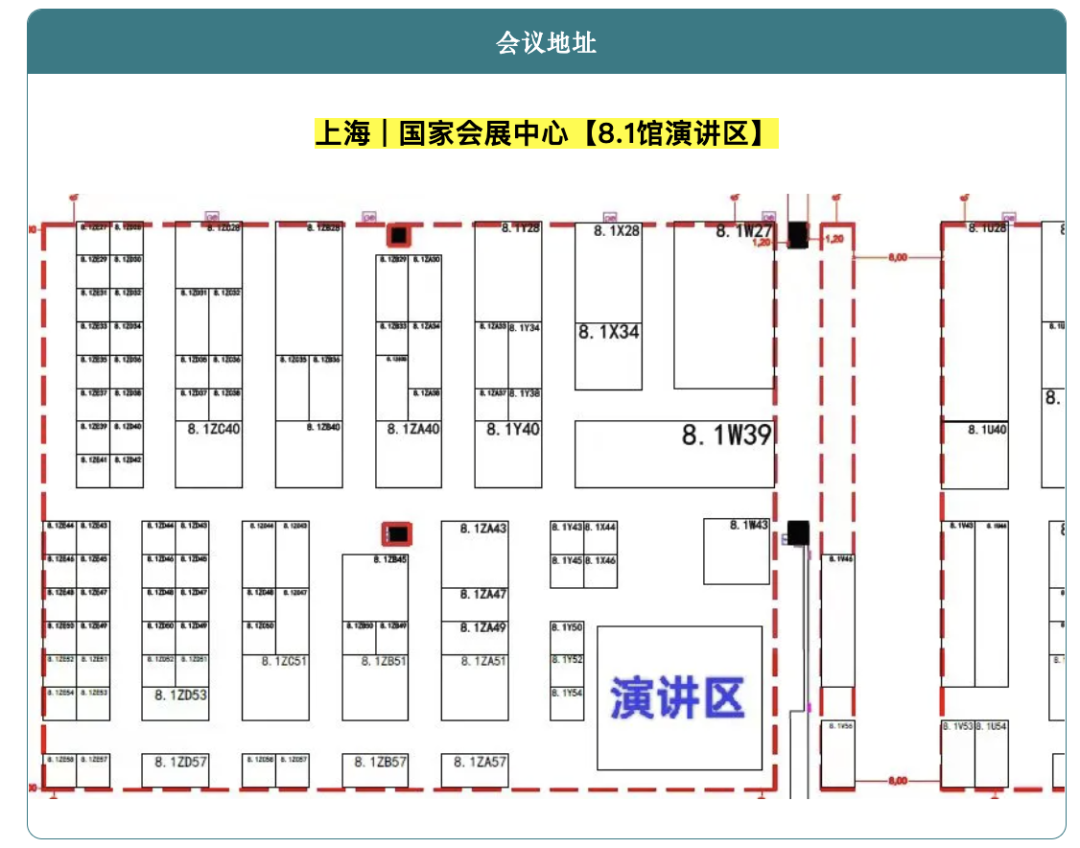
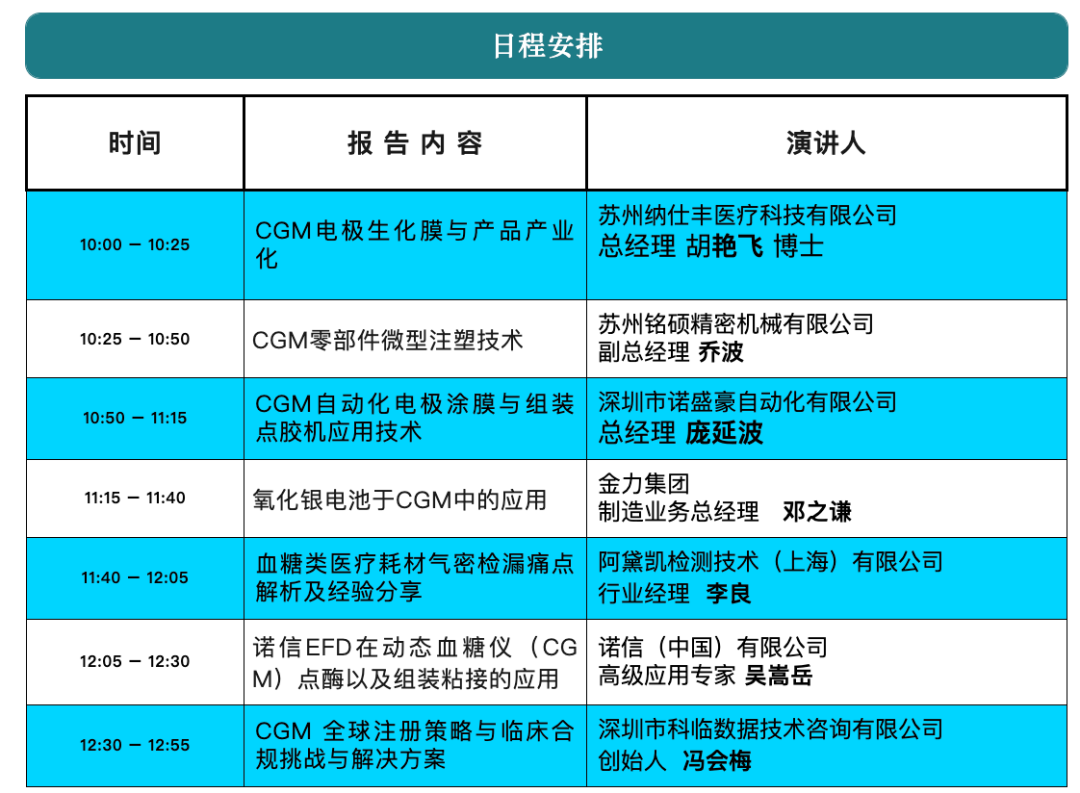
This technical seminar will be held on the morning of April 10 in the presentation area of Hall 8.1 at the National Exhibition and Convention Center. The seminar will bring together upstream and downstream companies across the CGM industry chain, including enterprises involved in electrode materials, biochemical membranes, micro-injection molding, automation equipment, battery technology, testing solutions, and adhesive technologies, to jointly explore CGM technology development trends and paths to industrialization.
In recent years, as the number of patients with diabetes continues to rise, demand for glucose monitoring has continued to expand. According to the International Diabetes Federation (IDF), the global number of people with diabetes has exceeded 500 million, with China ranking first in patient population worldwide. As a next-generation glucose monitoring technology, Continuous Glucose Monitoring (CGM), compared with traditional fingerstick blood sampling, can provide continuous, real-time, dynamic glucose data, significantly improving glucose management efficiency and patient adherence.
Driven by both technological progress and market demand, the global CGM market is expanding rapidly. Data show that the global CGM market grew from approximately USD 1.7 billion in 2015 to USD 5.7 billion in 2020, and is projected to reach USD 36.5 billion by 2030. Although the China CGM market started relatively late, it has developed at an exceptionally rapid pace and offers enormous future market potential.
However, for CGM companies, taking a product from R&D to commercialization requires not only technological breakthroughs, but, more importantly, systematic planning of global registration and clinical compliance strategies.Different countries and regions have significant differences in medical device regulations, clinical evaluation, market approval, and post-market surveillance. How to build an evidence package that meets international regulatory requirements during the R&D stage, and how to efficiently design registration pathways under regulatory systems such as the EU MDR, the U.S. FDA, and China’s NMPA, have become key issues in companies’ international market access strategies.