← Back to English Insights
Medical device regulatory insight · 2026-03-13
Shenzhen Clinsota to Exhibit at CMEF
2026|Presentation on Global CGM Registration Strategies and Clinical Compliance Solutions

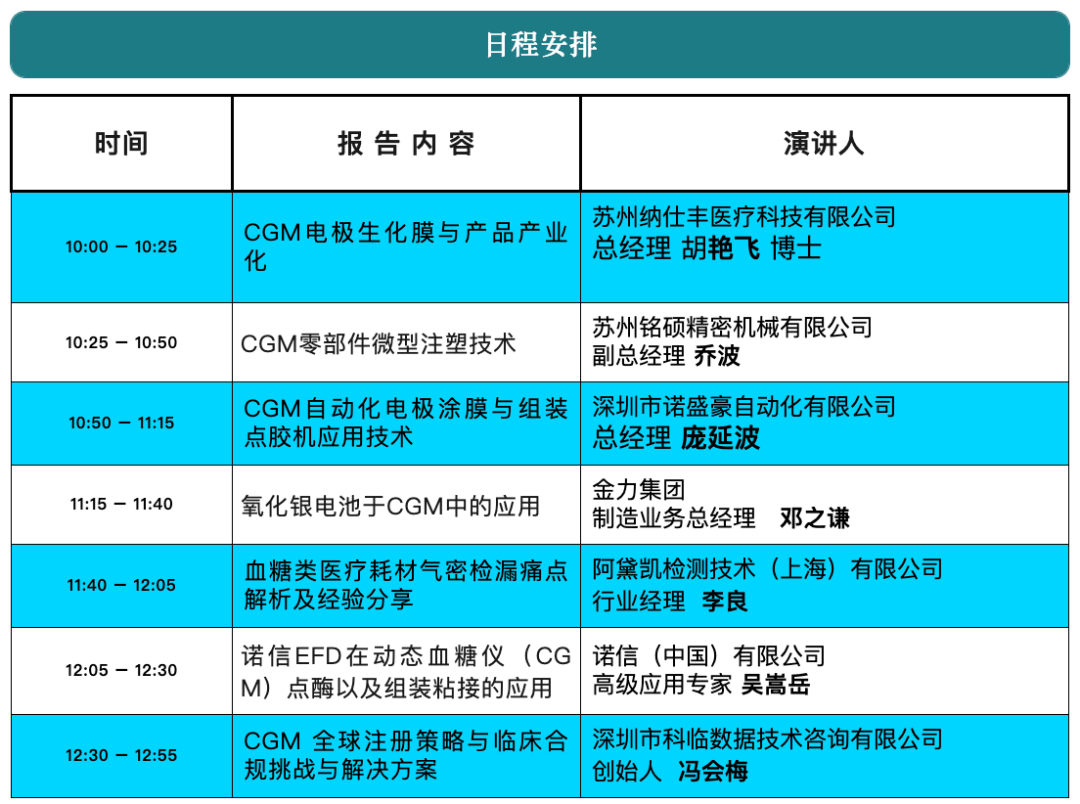
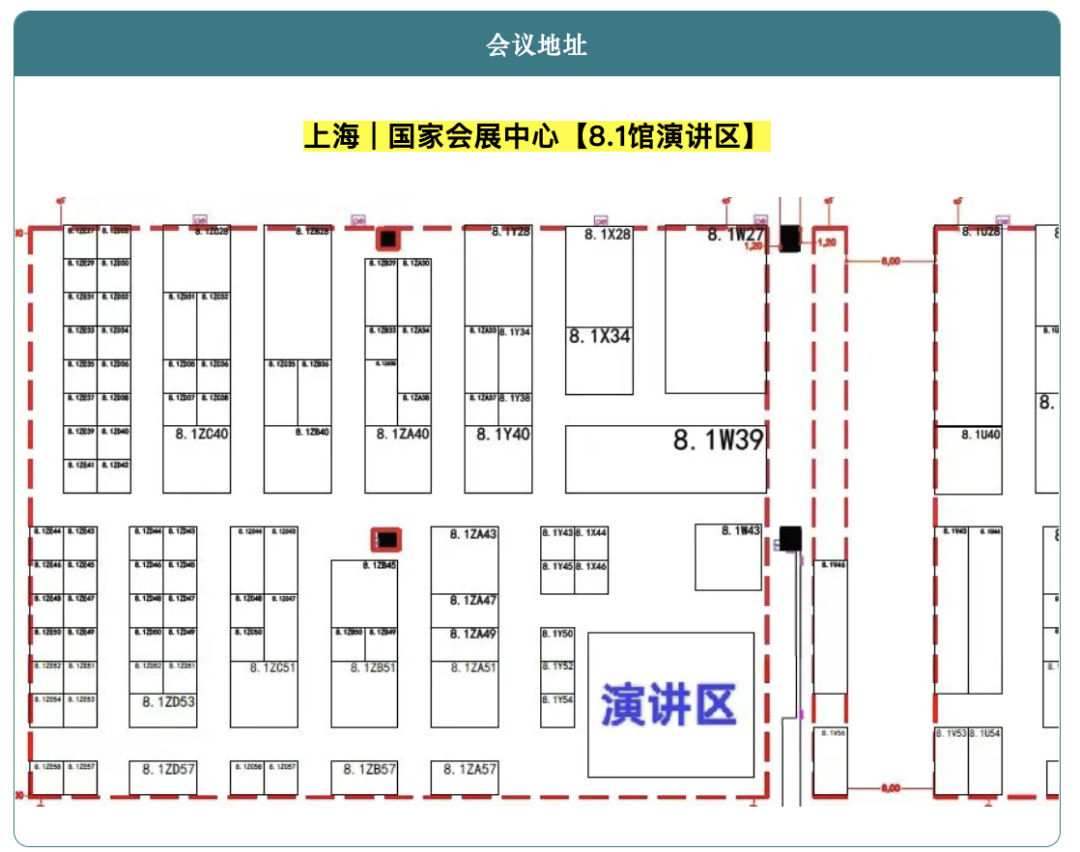
On the morning of April 10, 2026, Shenzhen Clinsota Technology Consulting Co., Ltd. will appear at the CMEF 2026 Spring Exhibition held at the National Exhibition and Convention Center (Shanghai), and has been invited to participate in the “2026 3rd Continuous Glucose Monitoring (CGM) Technology Seminar” to deliver a featured presentation: “Global Registration Strategies for CGM and Clinical Compliance Challenges and Solutions.”This technical seminar will be held on the morning of April 10 in the Presentation Area of Hall 8.1 at the National Exhibition and Convention Center. The meeting brings together upstream and downstream companies across the CGM value chain, including enterprises focused on electrode materials, biochemical membranes, micro-injection molding, automation equipment, battery technology, testing solutions, and adhesive technologies, to jointly discuss CGM technology trends and pathways to industrialization.Driven by both technological advances and market demand, the global CGM market is expanding rapidly. Data show that the global CGM market grew from approximately USD 1.7 billion in 2015 to USD 5.7 billion in 2020, and is projected to reach USD 36.5 billion by 2030. Although China’s CGM market started relatively late, it is developing at an exceptional pace and offers substantial future market potential.However, for CGM companies, bringing products from R&D to commercialization requires more than technological breakthroughs; even more critical is the systematic planning of global registration and clinical compliance strategies. Significant differences exist across countries and regions in medical device regulations, clinical evaluation, premarket approval, and post-market regulatory requirements.How to build an evidence framework aligned with international regulatory requirements during the R&D stage, and how to efficiently design registration pathways under regulatory systems such as EU MDR, the U.S. FDA, and China NMPA, have become key priorities for companies pursuing international market expansion.At this seminar,Feng Huimei, Founder and CEO of Shenzhen Clinsota Technology Consulting Co., Ltd., will deliver a special presentation on “Global Registration Strategies for CGM and Clinical Compliance Challenges and Solutions”. Drawing on many years of practical experience in international registration and clinical evaluation, the presentation will focus on common compliance challenges encountered during global market access for CGM products, including:• Overall planning and strategic positioning of registration pathways for CGM products under the EU MDR, FDA, and NMPA systems• Clinical Evaluation Report (CER) and clinical evidence development strategies• GAP analysis on how domestic clinical trial data can meet EU clinical requirements, enabling dual China-EU submission to reduce cost and accelerate timelines• Post-market Clinical Follow-up (PMCF) and full lifecycle managementShenzhen Clinsota Technology Consulting Co., Ltd. has long specialized in global medical device registration and clinical compliance services, and is committed to providing medical device companies with full-lifecycle compliance solutions from the R&D stage through post-market activities. The company’s team has extensive practical experience with EU MDR, FDA, and China NMPA registration systems, with particular depth in clinical evaluation, clinical evidence strategy, and international registration pathway design.During CMEF,Clinsota will also engage with industry partners on global registration planning for CGM products, clinical strategy design, and AI-driven automated clinical evaluation solutions. Built on a multi-agent architecture, this clinical compliance engine automatically performs intelligent retrieval and in-depth analysis of CER literature and establishes a dynamic automated update mechanism for post-market PMS/PMCF data, enabling end-to-end compliance management and driving exponential gains in global registration efficiency. Together, we aim to help Chinese medical device companies accelerate entry into global markets.We sincerely invite companies across the CGM value chain, R&D teams, and leaders responsible for medical device registration and clinical affairs to visit and connect with us on site.