← Back to English Insights
Medical device regulatory insight · 2026-04-25
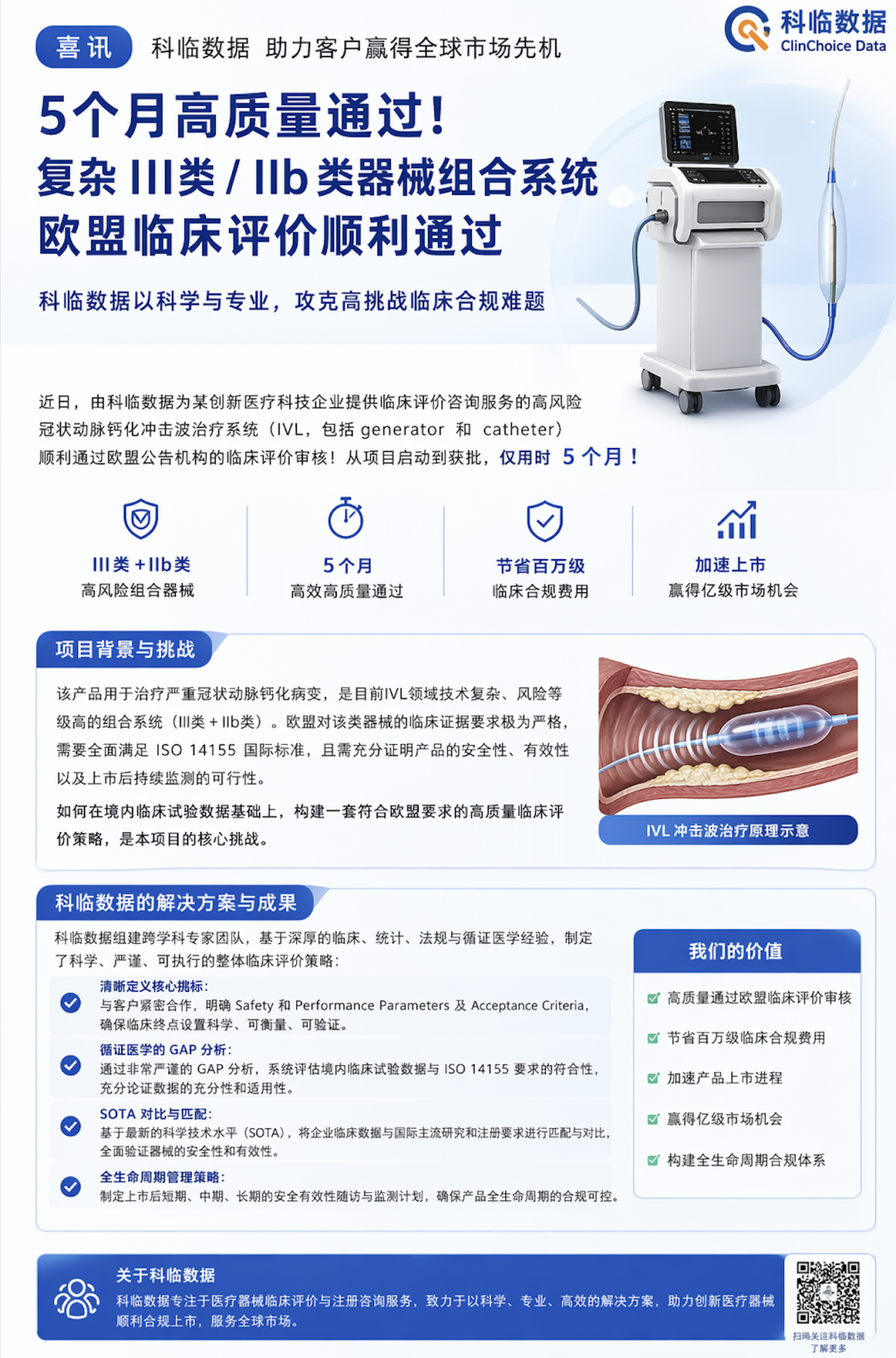
[News] Clinsota enabled a company to achieve EU clinical compliance for Class III and Class IIb high-risk combination products in just 5 months
Amid the challenging EU MDR landscape for medical-device clinical compliance, Kelin Data has good news to share: a complex coronary calcification intravascular lithotripsy (IVL) combination device that we supported successfully passed the EU pre-market clinical evaluation and compliance review for t
![[News] Clinsota enabled a company to achieve EU clinical compliance for Class III and Class IIb high-risk combination products in just 5 months](https://clinsota.com/content/images/2026/04/640-186.jpg)
At a time when EU MDR clinical compliance for medical devices remains highly challenging, Clinsota has good news worth sharing: one complexcoronary calcification intravascular lithotripsy (IVL) combination devicein just5 months, successfully completingthe EU pre-market Clinical Evaluation and conformity assessment for two high-risk combination products, one Class III and one Class IIb. This achievement not only helped the company savemillions in clinical compliance costs, but also created a market opportunity for rapid commercialization and entry into a market worth hundreds of millions.A complex, high-risk IVL combination system
This product is anintravascular lithotripsy system for treating severe calcified coronary lesions, comprising a unit that generates high-energy shockwavesgeneratorand a device that delivers energy and comes into contact with the patient's vasculaturecatheter. Unlike conventional rotational atherectomy, the IVL system uses tunedacoustic pressure wavesto create microfractures in calcified plaque and increase vascular compliance, thereby facilitating stent expansion by the physician. The shockwave energy passes through healthy tissue and creates fractures only within calcified plaque.This combination system involves two different risk classes: the generator is an active therapeutic device. Under Rules 1 and 9 of Annex VIII to the EU Medical Device Regulation (MDR), the shockwave generator is classified asClass IIb.The catheter is an interventional cardiovascular device. EU MDR guidance states that cardiovascular catheters which directly contact the heart or central circulatory system and are intended to control, monitor, or correct cardiac defects areClass III. Therefore, this is a high-risk combination system consisting of a Class III catheter and a Class IIb generator.High risk means very stringent requirements for clinical safety and performance, while also requiring the combination product to meet compliance requirements under different regulatory frameworks—precisely the challenge faced by the company.Clinsota’s clinical team applied a rigorous clinical evaluation strategy: meeting international standards with domestic data
For this high-risk combination device, the Clinsota team designed a “right-first-time” clinical evaluation strategy. Because the system had already completed a domestic registration clinical trial and generated high-quality data, we decided to make full use of these domestic data and demonstrate compliance with EU requirements through rigorous closed-loop evidence analysis based on evidence-based medicine.Evaluation framework designed in accordance with international standards. ISO 14155 is the international Good Clinical Practice standard for medical device clinical investigations, intended to protect subject rights, ensure scientific rigor, and define the responsibilities of sponsors and investigators [5]. In our evaluation framework, we followed this standard’s requirements for study design, conduct, recording, and reporting. We also performed a comprehensive GAP analysis. We compared the domestic clinical trial data point by point against the requirements of ISO 14155 and the EU MDR, identified gaps, and proposed supplementary measures, such as improving subject follow-up and adding risk assessment reports. Through systematic gap analysis, we ensured the completeness of the evidence while avoiding duplicate studies and saving substantial time and cost.Justification that domestic data meet international requirementsDuring the evidence evaluation process, we fully substantiated that the design and conduct of the domestic trial complied with international Good Clinical Practice, including protection of subject rights, randomization and blinding design, and data collection and monitoring. Through standardized data processing and statistical analysis, we ensured that the results were scientifically robust and reliable, enabling the domestic data to be used directly in the EU regulatory submission.SOTA analysis and safety and performance validationClinical evaluation requires not only data, but also analysis of those data in the proper context. The EU MDR explicitly requires“State of the Art (SOTA)”analysis, using currently accepted best practice as the comparator benchmark to assess the device’s advantages and limitations [6]. The Clinsota team conducted a comprehensive literature search and comparative device analysis, established an SOTA dossier for this class of therapy, and on that basis defined the clinical evaluation objectives:- Define safety and performance parameters:In alignment with EU regulations and the SOTA, we defined key indicators such as improvement in vessel compliance, effectiveness of vascular calcium modification, stent expansion performance, procedural success rate, and complication rate; for example, 30-day MACE ≤ 10%;
- Set acceptance criteria: acceptable risk thresholds were defined based on published data and clinical guidelines for similar treatment modalities;
- Comparative analysis of safety and effectiveness versus traditional alternatives, such as Rotational Atherectomy, Orbital Atherectomy, cutting/scoring balloons, and high-pressure non-compliant balloons;
- Matching and comparative analysis of clinical data against SOTA benchmarks: by matching and comparing the company’s own clinical data against SOTA benchmarks, we demonstrated that the system is equivalent or superior to existing treatment options in safety and performance;
- Lifecycle management strategy: in addition to pre-market studies, we developed short-, medium-, and long-term post-market clinical follow-up plans to ensure ongoing monitoring of safety and effectiveness.
Through this work, we verified the system’s short- and long-term safety and effectiveness for patients, meeting the EU’s stringent requirements for high-risk devices.Reduce costs, accelerate market access, and win the market
The traditional approach would be to conduct another overseas clinical trial, which is not only time-consuming and costly, but can also delay market opportunities. By fully leveraging existing high-quality data and strictly adhering to international standards, our strategy avoided duplicate clinical trials and saved the companymillions in clinical compliance costs and shortened the time to market to 5 months. This not only significantly reduced the company's financial burden, but also enabled the product to reach the market within the optimal market window, securing a market opportunity worth hundreds of millions of RMB.Clinsota: A trusted global compliance partner for medical devices, combining expertise with practical execution
This case once again demonstrates:- Expertise — We specialize in EU MDR and international standards, helping remove clinical compliance barriers for high-risk combination products.
- Efficiency — Within 5 months, we completed the full process from gap analysis and data review to supplemental studies and regulatory submission. Achieving clinical compliance for two high-risk combination products—Class III and Class IIb—within five months is exceptionally rare.
- Practicality — We communicate with clients in clear, accessible language, helping companies understand regulatory requirements and technical details; at the same time, we work closely on the ground with clinical institutions and corporate R&D teams.
- Trusted for the long term — We focus not only on pre-market compliance, but also on post-market surveillance and risk management, supporting products throughout their full lifecycle.
Looking ahead, Clinsota will continue to support more innovative medical devices with a pragmatic and professional approach. If you are also facing challenges in clinical evaluation and compliance, feel free to contact us,and let us help bring good products to market faster and more cost-effectively.
![[News] Clinsota enabled a company to achieve EU clinical compliance for Class III and Class IIb high-risk combination products in just 5 months](https://clinsota.com/content/images/2026/04/640-186.jpg)