I. What the expert recommendations are really trying to address is whether it “can be used for medical decision-making”
If you read the full document, you will find that the expert recommendations consistently revolve around one core question:
Under what conditions can data collected by wearable devices be incorporated into clinical judgment and medical decision-making?
To address this, the expert panel adopted a graded recommendation approach,
classifying different application scenarios into “Strongly Recommended, Recommended, May Be Considered, Not Recommended” as four levels:

This grading is not a conceptual assessment; it directly corresponds to the sufficiency of evidence, controllability of risk, and application maturity.
From a CRO perspective, this grading system is essentially the “A guidepost”:
It tells researchers which directions are suitable for registration studies,
which are better suited to real-world exploration,
and which should be avoided for the time being to prevent wasted resources.
II. Areas with relativelymature application evidence:where clinical studies can proceed more steadily
In the expert recommendations, several application categories are mentioned repeatedly, with relatively clear support:
First, arrhythmia screening and monitoring represented by atrial fibrillation
Experts believe that, in specific populations, wearable devices based on photoplethysmography or ECG signals already have relatively reliable screening value. At the same time, however, they clearly emphasize the boundary between “screening” and “diagnosis”—any abnormal finding still requires confirmation by a standard ECG examination.
Second, patch-based long-term ECG monitoring
Compared with traditional Holter monitoring, patch devices offer practical advantages in wearing comfort and monitoring duration. Expert recommendations suggest that, provided the use scenarios and comparator methods are clearly defined, such devices can serve as a supplement to existing monitoring modalities. This also provides a relatively clear positioning for related clinical studies.
Third, blood pressure monitoring conducted under standardized conditions
Experts gave relatively positive evaluations of cuff-based blood pressure monitoring, while also emphasizing the importance of measurement posture, use procedures, and the applicable population. This means that clinical studies must focus not only on device performance, but also on strict control of conditions of use.
III. Scenarios that still requirecaution: when evidence is insufficient, boundaries are especially important
Corresponding to the directions above, the expert recommendations also clearly identify some applications that still require cautious consideration.
For example,cuffless blood pressure measurement、energy expenditure assessment、and functions such as sleep staging, while continuing to improve technically, still have limitations in clinical accuracy and stability,and still lack an evidence base sufficient to support medical decision-making。
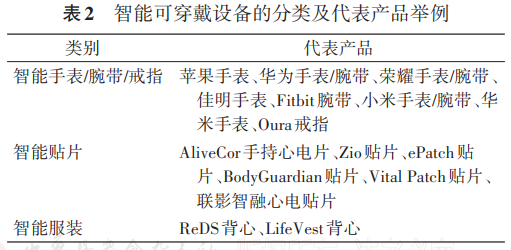
Therefore, it is better suited to health management or long-term data accumulation rather than direct entry into the medical regulatory submission pathway.
Based on CRO experience, overemphasizing medical attributes too early in these scenarios often creates unnecessary resistance during ethics review, protocol assessment, or regulatory communication.
Before the evidence base is sufficiently mature, carefully calibrating Intended Claims is the more prudent choice.
what gold standard should be used to confirm an abnormal alert;
how the confirmed result should be documented and communicated;
whether further referral or intervention is required.
repeated discussion of product positioning and intended use;
sound design of clinical endpoints, comparator approach, and sample size; and early identification and documented management of potential risks.
Conclusion: Let consensus be the starting point, not the barrier
The publication of these expert recommendations is not intended to impose new restrictions on the industry, but to provide a practical assessment framework for complex and diverse cardiovascular wearable applications.
As a CRO working alongside frontline clinical research teams, Clinsota plays a more important role in helping companies clarify the research pathway and strengthen the foundational work based on this consensus.
In the wearable cardiovascular field:
carefully balancing “what can be demonstrated, Intended Claims, and evidence-based validation”, may be what enables innovation to go further.
These expert recommendations are worth understandingand internalizing,and we also welcome further discussion with you at any time.
