In one sentence, here is the conclusion:
👉 Medical device registration documents,are not “submission materials”; they are “legal documents”。
👉 Falsification does not meanrejection; it meansregistration certificate revocation。
Over the past two years, the regulatory stance has become unmistakably clear.

Product: Disposable endotracheal tube
Registration Certificate No.: 苏械注准20182081539
Outcome:Medical device registration certificate revoked, illegal gains confiscated,no medical device licensing applications accepted for 10 years
Basis:Regulations on the Supervision and Administration of Medical DevicesArticle 83, paragraph 1 (with reference to the Administrative Licensing Law)

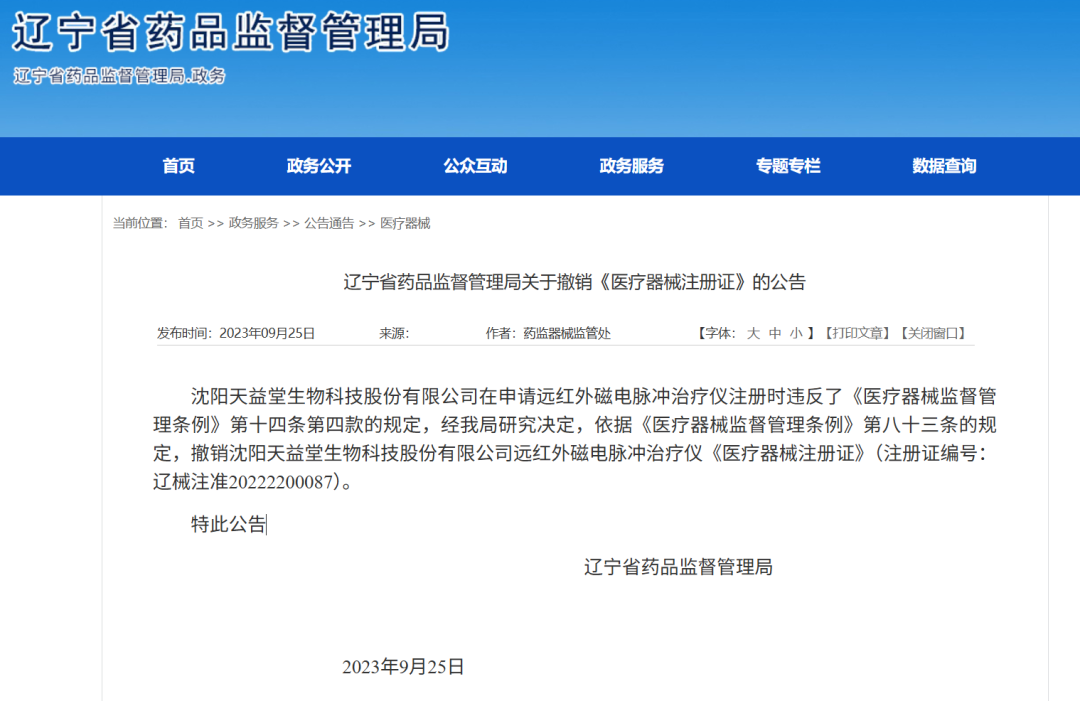
Product: Far-infrared magnetoelectric pulse therapy device
Registration Certificate No.: 辽械注准20222200087
Outcome:Medical device registration certificate revoked
Reason: the registration certificate application violated theprovisions of Article 14, paragraph 4, of the Regulations on the Supervision and Administration of Medical Devices
What you need to see clearly is not the “cases,” but the trend
These three cases show severalhighly consistent signals:
❌ Not a “quality issue”
❌ Not an “adverse event”
❌ Not a “clinical failure”
But rather—
⚠️ Untruthful registration documents / submission of false information
Regulators have repeatedly shown through enforcement that:
Once you cross the line into “false submission,” there is no discussion of room for correction; eligibility for approval is denied outright。
What, in essence, are registration documents?
All medical-device professionals should remember one sentence:
The registration dossier you submit is a formal legal representation made to the national regulatory authority.
Not PPT
Not an internal report
Not “close enough”
But rather:
You can be held accountable
It can be investigated retroactively
Administrative approval can be revoked directly
By function: an action checklist you can implement today
For medical-device business owners
Make “registration dossier compliance” a red-line item: assign resources, time, and clear accountability
In one sentence:Do not let performance KPIs push your team into compliance incidents!!!
For Registration/Regulatory Affairs (RA)
Create a “key statements checklist”: every commitment written into the submission dossier must besupported by evidence
Treat Article 83 as a bottom-line provisionTraining and sign-off
For Quality Assurance (QA)
Focus on: record authenticity, supplier control, outsourced processes, and data integrity
Ensure the “review dossier” is consistent with actual QMS practice!!!Do not run two systems
For R&D
Do not submit PPT-style conclusions only. For verification, validation, and risk managementcomplete the evidence chain
For every change, ask:Could this affect a commitment made in the submission dossier?
For Clinical/Medical Affairs (CM/MA)
Clinical evaluation is not a writing contest; it is a test of evidentiary logic
For equivalent device selection, difference analysis, and applicability justification, the more critical they are, the less room there is for ambiguity
Final reminder (please keep the original wording)
In a medical device registration dossier, not a single page can be falsified.
Because the cost is not “redoing the dossier,” but—revocation of the registration certificate.
If your company is currently:
pursuing registration
supplementing registration materials
preparing a change submission
or has already been on the market for many years
👉 now is exactly the time to look back and verify the authenticity of your registration documents.

